We rarely need to imagine the situation in the summers when the temperature crosses the 45-degree mark and makes life miserable. Heat strokes in certain parts of India cause many deaths and skin-related diseases see multifold increases. When 45° to 50° degree temperature takes the life out of us humans and other living beings; What if the temperature on the Earth touches the range of 5,227 to 5,727 °C?
Well, that is the actual radiation the Sun emanates on a daily basis to the Earth. Then what makes survival possible for living beings on Earth? Ozone Layer.
What is an Ozone Layer?
Two French physicists discover in the year 1913 that Sun sends out radiation around 5000 °C when it reaches the Earth, which can make life non-existential on the Earth. Charles Fabry and Henri Buisson established the fact that there is something that absorbs almost all the mid-range radiation of the Sun but failed to find out the exact element that absorbs the ultraviolet rays coming from the Sun.
G.M.B Dobson, during his research, established the fact that the element that absorbs almost 97 to 99% of the Sun’s radiation is Ozone. He developed a Dobsonmeter to measure the effectiveness of the Ozone layer from the ground. In his research, he established that the missing radiation that is supposed to reach to the Earth matches that of the chemical known as Ozone, which covers the entire Earth and protects human life from the fury of the Sun.
The Distribution of the Ozone Layer
The thickness of the thinness of Ozone layer is not consistent and is mainly dependent on the greenhouse gases as well the season. In its usual form, the Ozone layer is thin around The Equator and thick near the poles. During the summers you can measure the Ozone layer being thin, while in the winters, it is thick. Besides, solar intensity plays a critical role in deciding the effectiveness of the Ozone Layer.
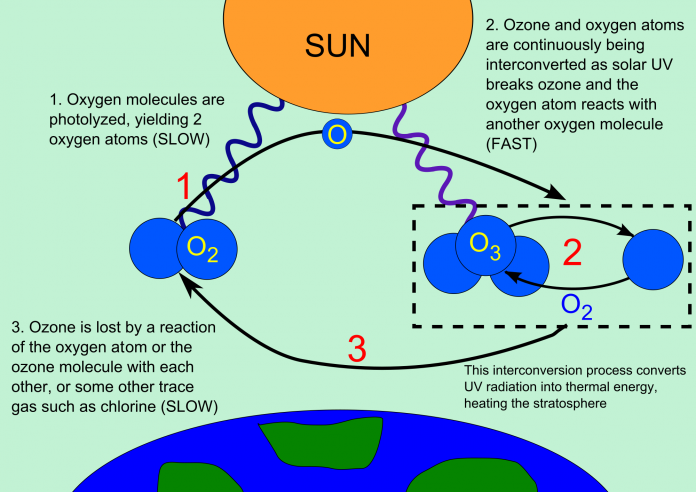
The Ozone Structure
Science sometimes throws unimaginable surprises. Ozone is a structure that is nothing less than an ultraviolet layer similar to the Sun’s UV rays, which is made of Oxygen only. While water consists of two Oxygen atoms, Ozone is made of three Oxygen atoms.
Effect Of Ozone Layer Depletion On Plants
UVB radiation has a direct impact on plants that alters their physiological and developmental processes. Despite the fact that plants have the ability to adapt to increased UVB levels, such radiation is damaging to the foliage. UVB radiation has the potential to cause indirect changes in plants. These changes are often more harmful than the direct impact of UVBs on plants. The modifications could include changes in plant form, nutrient distribution within the plant, developmental phase timing, and secondary metabolism.
Effects Of Ozone Layer Depletion On Marine Life
Scientists discovered that ozone layer depletion reduces the production of phytoplankton, which are the world’s largest oxygen producers. Furthermore, UV radiation has been shown to harm the developing stages of fish, crab, shrimp, amphibians, and other marine life.
Montreal Protocol
- The Montreal Protocol is an environmental agreement that controls the production and usage of Ozone Depleting Substances (ODS) that is resulting in the thinning of the ozone layer. The protocol was signed on September 16, 1987.
- The ratifiers of the Montreal Protocol are the 197 member states of the UN along with Niue, the Cook Islands, the Holy See and the European Union. The international treaty is the first of its that has been ratified universally in UN history.
- The Multilateral Fund for the Implementation of the Montreal Protocol (1991) is to provide financial as well as technical assistance to developing member countries to comply with the control measures of the Protocol. The only criteria to meet is that the annual per capita consumption and production of ODS for such country parties should be less than 0.3 kg.
- In countries like the US, chlorofluorocarbons and halons have been phased out. In India, various ODSs like CFCs, carbon tetrachloride, halons, methyl bromide have been phased out for controlled uses keeping the Montreal Protocol into consideration.
Why Ozone Layer?
Thinking of life on the Earth at the temperature of 5000 °C is an unthinkable thing. This fact itself explains the importance of the Ozone layer. Radiation that comes out of the Sun would not only destroy the plants and trees but would also kill the living beings from the Earth. Besides, if all the ice will melt in a day or two with such a high temperature, life on the Earth can never be possible. Even a one percent decrease in Ozone could affect the life severely on the Earth. Therefore, we must ensure that we do not eliminate harmful gases and preserve the environment as much as we should.
Frequently Asked Questions:
What is Ozone Layer Depletion?
Ozone depletion is the thinning of the ozone layer present in the stratosphere or upper atmosphere (9-18 miles/15-30km above the earth’s surface). Ozone layer depletion is caused by Ozone Depleting Substances (ODS) such as CFCs, hydrochlorofluorocarbons, halons, etc.
What are the Effects of Ozone Layer Depletion on Human Health?
With the depletion of the ozone layer, the UVB, or the UV light from the sun reaches the atmosphere and affects living beings in a major way. It damages DNA and causes melanoma and other types of cancer and skin diseases in humans. The ozone layer depletion can also lead to eye diseases like cataracts, snow blindness, and a weakened immune system. Moreover, it also causes harm to crops and marine life.
What Is the Current State of the Ozone Layer?
There are ozone holes both in the northern (Arctic) and the southern (Antarctica) hemispheres. The ozone over Antarctica is the larger one. In 2021, the ozone hole had a maximum of 24.8 million square kilometers. In 2022, a new ozone hole has been discovered above the tropics that is 7 times larger than the one over Antarctica.
Meanwhile, the ODSs have fallen by 26% from their high value in the 1990s (as per the 2022 Ozone Depleting Gas Index). This progress over 30 years has been achieved due to the control put on the reduction of the ODSs under the Montreal Protocol.
What Is the Importance of Ozone Layer?
The ozone layer helps in protecting the earth by preventing harmful radiation from coming from the sun. It also blocks the more dangerous UV light called UVB coming from the star. Without the ozone layer, life would suffer on earth or probably not exist.
What Causes Ozone Depletion?
Ozone depletion is caused by Ozone Depleting Substances (ODS) like Chlorofluorocarbons (CFCs), Halons, Carbon tetrachloride, Methyl Chloroform, Methyl bromide, and Hydrofluorocarbons. These chemicals are present in our everyday items of use like refrigerators, AC, deodorants, dry-cleaning agents, etc. These ODS react with high ultraviolet radiation to release chlorine and bromine atoms that result in the thinning of the ozone layer. One chlorine atom can destroy 100,000 ozone molecules. Only 1-2% of ozone depletion is caused by natural causes like sun spots, stratospheric winds, etc.
What is the Ozone Layer Made Of?
The ozone layer is made up of three atoms of oxygen, also called O3. It is formed by the reaction between oxides of nitrogen and hydrocarbons.
Which Day Celebrates World Ozone Day?
September 16 is celebrated as World Ozone Day every year. It is also known as International Ozone Day or International Day for the Preservation of the Ozone Layer. On this World Ozone Day, awareness is created about the ozone layer and how we can protect it by reducing things that result in its depletion. Students are educated, seminars are held and events are organized to provide more information about the ozone layer.
Who First Discovered the Ozone Layer?
The ozone layer was first discovered by French physicists Charles Fabry and Henri Buisson in the year 1913. The discovery was later developed by British meteorologist G.M.B Dobson who developed a Dobsonmeter to measure the ozone from the earth’s surface.
What is the Theme of World Ozone Day 2022?
World Ozone Day is observed on September 16 every year. The theme for 2022 World Ozone Day is Montreal Protocol@35: Global Cooperation Protecting Life on Earth. The theme recognizes the impact created by the Montreal Protocol on climate change. It aims to enhance collaboration, partnerships, and global cooperation to address pressing issues regarding the climate. It will also ensure that steps are taken to protect future generations from the adverse effects of climate change.
The Final Thought!
At a global level, countries have come forward and taken necessary steps to phase out the production and usage of Ozone Depleting Substances (ODS). We as individuals bear equal responsibility for reducing, if not eliminating, the use of items that contribute to the thinning of the ozone layer. If not today, the future generations will suffer solely because we chose to ignore the pressing issues that require our attention. Only by taking the necessary steps will we be able to save our mother earth from further harm.
Have you made any environmental contributions? We’re excited to hear about it. Please leave your thoughts in the comments section below.